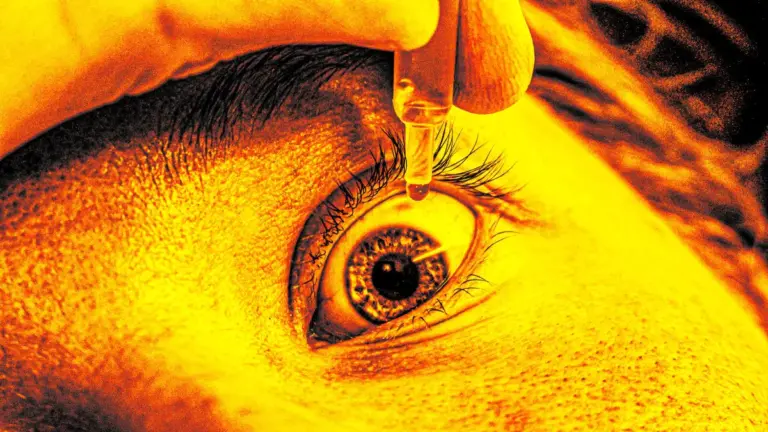
K.C. Pharmaceuticals recalled more than 3.1 million bottles of lubricating eye drops on March 3, 2026. The recall stems from sterility issues that could endanger consumer health.
Eight products are included in the recall, such as Dry Eye Relief Eye Drops and Artificial Tears Sterile Lubricant Eye Drops. These products were sold under various names at major retailers, including Walgreens, CVS, and Kroger.
The expiration dates for the recalled eye drops range from April 30, 2026, to October 31, 2026. As of early April 2026, no infections linked to these eye drops have been reported.
The FDA has previously identified sterility issues at K.C. Pharmaceuticals. In 2023, the agency linked contaminated eye drops to severe infections affecting 81 people, resulting in multiple recalls.
In that outbreak, four individuals experienced vision loss and four others had their eyeballs removed due to infections. Additionally, four deaths were reported in connection with contaminated eye drops.
The FDA has limited personnel and focuses inspections on higher-risk products. Before 2023, it inspected over-the-counter eye drop manufacturers only a few times.
In response to the current recall, a clinical pharmacologist stated, “Using nonsterile eye drops that harbor bacteria and fungus can cause eye infections.” This highlights the potential dangers associated with using these products.
If consumers find their product has been recalled, they should stop using it immediately and return it to the store for a refund.